Fintech
NanoViricides, Inc. Raises $11.5 Million Through Public Offering After Positive Test Results For COVID-19 Drug

The Company’s successful fundraising and inclusion in the Russell Microcap(R) Index comes after a series of promising test results for its COVID-19 drug candidates
Shelton, Connecticut–(Newsfile Corp. – July 23, 2020) – NanoViricides, Inc. (NYSE American: NNVC) (the “Company”) is a development stage, nano-biopharmaceutical Company, with proprietary research focused on countering viral diseases. The Company’s lead drug candidate, a topical cream for shingles, has been at the cusp of the IND (Investigational New Drug) application before heading for Phase 1 of human trials. Its research team has also been working on the development of a drug to treat the SARS-CoV-2 virus since the beginning of 2020 and has shown visible progress with positive results in the animal model.
Read Full Article Here: http://smallcapsdaily.com/excellent-safety-data-for-covid-19-drug-candidate-plus-15-million-in-cash-put-nanoviricides-in-the-drivers-seat/
The NanoViricides management announced the completion of a capital raise to the tune of $11.5 million through an underwritten public offering. The Company issued 1,369,863 shares and a fully exercised underwriters’ over-allotment option of 205,479 additional shares of NanoViricides’ common stock at the public offering price of $7.30 per share. This issue took place at an approximate discount of 17% over the stock’s closing price in the previous trading session and was underwritten by Kingswood Capital Markets, a division of Benchmark Investments, Inc. Kingswood Capital acted as sole bookrunner for the public offering. The overall net proceeds with the Company after the pay out of commission and other transaction fees is approximately $10.53 million, before deducting the Company’s legal and accounting expenses related to the offering. It is worth highlighting that prior to this offering, the management reported cash and cash equivalents worth $6.11 million in its most recent quarterly result. Currently, the Company has over $15 million in cash available for funding its research and meeting its working capital requirements. Notably, the Company had been added to the Russell Microcap® Index as of June 29, 2020.
Prior to the public offering, NanoViricides featured in the news for significant progress made on its COVID-19 drug candidate. The Company’s proprietary nanoviricide® technology, which works by trapping virus particles the same way as a Venus-fly-trap captures and consumes insects, has wide-ranging antiviral applications and is being used in drug development against many different viruses including COVID-19, shingles, herpes, HIV, and others. With respect to COVID-19, the Company has developed broad-spectrum anti-coronavirus drug candidates and carried out initial tests using cell culture assays as well as through an animal model. In its tests using cell culture assays of two different coronaviruses, namely hCoV-229E, and hCoV-NL63, the Company’s drug candidates have proven to be far more effective than favipiravir, a commonly used antiviral medication used for treating SARS-CoV-2. While the coronavirus samples used in the cell culture studies were not the SARS-CoV-2, they have related cellular receptors, namely APN which provides a rational basis to the research team. These tests were followed by the use of an animal model of the human coronavirus disease. The results were compared with remdesivir, the popular broad-spectrum antiviral medication developed by Gilead Sciences that has been approved as a treatment for COVID-19 patients. NanoViricides’ candidates showed superior results to remdesivir in the animal model. For the animal study, the researchers employed the hCoV-NL63, the same coronavirus used in the cell culture assays which uses the same cell receptor, ACE2, as does the SARS-CoV-2. While the hCoV-NL3 is a milder strain of the coronavirus, it acts as a suitable surrogate for the study that can be done in the Company’s BSL-2 lab. The Company would require BSL-3 or BSL-4 labs for carrying out similar studies using the actual SARS-CoV-2 strain for which the management is on the lookout for potential collaborations. The results of these tests were encouraging and prompted the Company to move ahead with safety and tolerability studies on its COVID-19 drug candidates.
As a part of its safety and tolerability tests, the Company’s research team tested three different drug candidates at three different dosage levels and vehicle control through separate groups of mice intravenously. Sixteen mice in each group, eight males and eight females, were administered one of the three drug candidates at one of the three dose levels, and additionally, one group was administered vehicle control, for seven days by daily tail-vein intravenous infusion in this blinded study with additional evaluations on the eighth day. This non-GLP safety and tolerability study was conducted under GLP-like conditions by AR BioSystems in Tampa, Florida. The results indicated that tested drug candidates were safe and well-tolerated with no clinical signs of immune or allergic reactions. The results showed no observable changes in any organs, including the large intestine or colon, on post mortem in gross histology. The only reportable changes were, in the high dosage groups of two of the three drug candidates tested, associated with the non-absorption of water, in the colon. This is consistent with the clinical observation of very loosened stools in the same groups. The positive result is expected to enable the Company to move forward with further development of its candidates and advancing to human trials.
Prior to advancing with human clinical trials, the Company’s management plans to conduct studies to carry out clinical candidate selection and possibly request a pre-IND meeting with the FDA for regulatory guidance. The management looks to engage in strong collaborations in the future and can also expect cash flows expected through licensing once its candidates for shingles as well as the SARS-CoV-2 start clearing the initial phases of human trials.
About NanoViricides
NanoViricides, Inc. (AMEX: NNVC) is a development stage Company that is creating special purpose nanomaterials for antiviral therapy. The Company’s novel nanoviricide® class of drug candidates are designed to specifically attack enveloped virus particles and to dismantle them. The Company is developing drugs against a number of viral diseases including H1N1 swine flu, H5N1 bird flu, seasonal Influenza, HIV, oral and genital Herpes, shingles and chickenpox, viral diseases of the eye including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue fever, and Ebola virus, among others. This press release contains forward-looking statements that reflect the Company’s current expectation regarding future events. Actual events could differ materially and substantially from those projected herein and depend on a number of factors. Certain statements in this release, and other written or oral statements made by NanoViricides, Inc. are “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company’s control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future. Important factors that could cause actual results to differ materially from the Company’s expectations include, but are not limited to, those factors that are disclosed under the heading “Risk Factors” and elsewhere in documents filed by the Company from time to time with the United States Securities and Exchange Commission and other regulatory authorities. Although it is not possible to predict or identify all such factors, they may include the following: demonstration and proof of principle in preclinical trials that a nanoviricide is safe and effective; successful development of our product candidates; our ability to seek and obtain regulatory approvals, including with respect to the indications we are seeking; the successful commercialization of our product candidates; and market acceptance of our products. For more details on the Company, please visit http://www.nanoviricides.com/.
Forward-Looking Statement
This press release contains forward-looking statements that reflect the Company’s current expectation regarding future events. Actual events could differ materially and substantially from those projected herein and depend on a number of factors. Certain statements in this release, and other written or oral statements made by NanoViricides, Inc. are “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company’s control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future. Important factors that could cause actual results to differ materially from the company’s expectations include, but are not limited to, those factors that are disclosed under the heading “Risk Factors” and elsewhere in documents filed by the company from time to time with the United States Securities and Exchange Commission and other regulatory authorities. Although it is not possible to predict or identify all such factors, they may include the following: demonstration and proof of principle in preclinical trials that a nanoviricide is safe and effective; successful development of our product candidates; our ability to seek and obtain regulatory approvals, including with respect to the indications we are seeking; the successful commercialization of our product candidates; and market acceptance of our products. FDA refers to US Food and Drug Administration. IND application refers to “Investigational New Drug” application. CMC refers to “Chemistry, Manufacture, and Controls”.
Contact:
Smallcaps Daily
Email: [email protected]
Website: https://smallcapsdaily.com
Fintech
How to identify authenticity in crypto influencer channels

Modern brands stake on influencer marketing, with 76% of users making a purchase after seeing a product on social media.The cryptocurrency industry is no exception to this trend. However, promoting crypto products through influencer marketing can be particularly challenging. Crypto influencers pose a significant risk to a brand’s reputation and ROI due to rampant scams. Approximately 80% of channels provide fake statistics, including followers counts and engagement metrics. Additionally, this niche is characterized by high CPMs, which can increase the risk of financial loss for brands.
In this article Nadia Bubennnikova, Head of agency Famesters, will explore the most important things to look for in crypto channels to find the perfect match for influencer marketing collaborations.
-
Comments
There are several levels related to this point.
LEVEL 1
Analyze approximately 10 of the channel’s latest videos, looking through the comments to ensure they are not purchased from dubious sources. For example, such comments as “Yes sir, great video!”; “Thanks!”; “Love you man!”; “Quality content”, and others most certainly are bot-generated and should be avoided.
Just to compare:
LEVEL 2
Don’t rush to conclude that you’ve discovered the perfect crypto channel just because you’ve come across some logical comments that align with the video’s topic. This may seem controversial, but it’s important to dive deeper. When you encounter a channel with logical comments, ensure that they are unique and not duplicated under the description box. Some creators are smarter than just buying comments from the first link that Google shows you when you search “buy YouTube comments”. They generate topics, provide multiple examples, or upload lists of examples, all produced by AI. You can either manually review the comments or use a script to parse all the YouTube comments into an Excel file. Then, add a formula to highlight any duplicates.
LEVEL 3
It is also a must to check the names of the profiles that leave the comments: most of the bot-generated comments are easy to track: they will all have the usernames made of random symbols and numbers, random first and last name combinations, “Habibi”, etc. No profile pictures on all comments is also a red flag.
LEVEL 4
Another important factor to consider when assessing comment authenticity is the posting date. If all the comments were posted on the same day, it’s likely that the traffic was purchased.
2. Average views number per video
This is indeed one of the key metrics to consider when selecting an influencer for collaboration, regardless of the product type. What specific factors should we focus on?
First & foremost: the views dynamics on the channel. The most desirable type of YouTube channel in terms of views is one that maintains stable viewership across all of its videos. This stability serves as proof of an active and loyal audience genuinely interested in the creator’s content, unlike channels where views vary significantly from one video to another.
Many unauthentic crypto channels not only buy YouTube comments but also invest in increasing video views to create the impression of stability. So, what exactly should we look at in terms of views? Firstly, calculate the average number of views based on the ten latest videos. Then, compare this figure to the views of the most recent videos posted within the past week. If you notice that these new videos have nearly the same number of views as those posted a month or two ago, it’s a clear red flag. Typically, a YouTube channel experiences lower views on new videos, with the number increasing organically each day as the audience engages with the content. If you see a video posted just three days ago already garnering 30k views, matching the total views of older videos, it’s a sign of fraudulent traffic purchased to create the illusion of view stability.
3. Influencer’s channel statistics
The primary statistics of interest are region and demographic split, and sometimes the device types of the viewers.
LEVEL 1
When reviewing the shared statistics, the first step is to request a video screencast instead of a simple screenshot. This is because it takes more time to organically edit a video than a screenshot, making it harder to manipulate the statistics. If the creator refuses, step two (if only screenshots are provided) is to download them and check the file’s properties on your computer. Look for details such as whether it was created with Adobe Photoshop or the color profile, typically Adobe RGB, to determine if the screenshot has been edited.
LEVEL 2
After confirming the authenticity of the stats screenshot, it’s crucial to analyze the data. For instance, if you’re examining a channel conducted in Spanish with all videos filmed in the same language, it would raise concerns to find a significant audience from countries like India or Turkey. This discrepancy, where the audience doesn’t align with regions known for speaking the language, is a red flag.
If we’re considering an English-language crypto channel, it typically suggests an international audience, as English’s global use for quality educational content on niche topics like crypto. However, certain considerations apply. For instance, if an English-speaking channel shows a significant percentage of Polish viewers (15% to 30%) without any mention of the Polish language, it could indicate fake followers and views. However, if the channel’s creator is Polish, occasionally posts videos in Polish alongside English, and receives Polish comments, it’s important not to rush to conclusions.
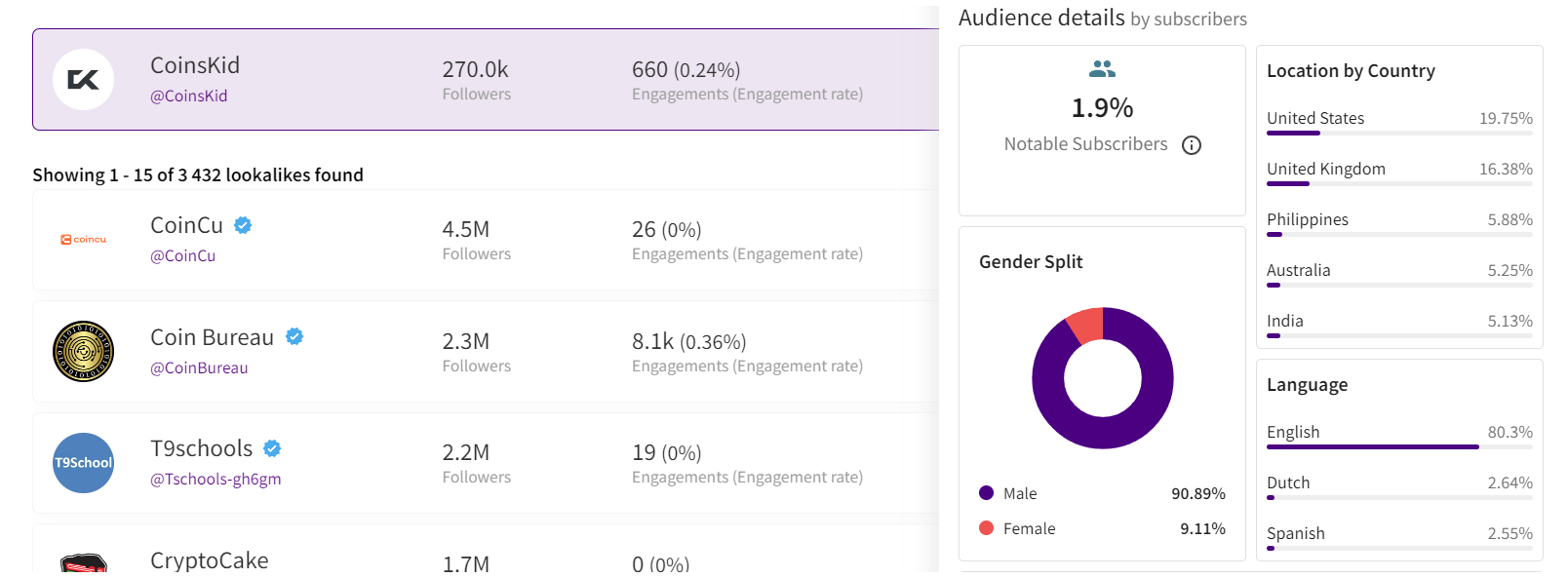 Example of statistics
Example of statistics
Wrapping up
These are the main factors to consider when selecting an influencer to promote your crypto product. Once you’ve launched the campaign, there are also some markers to show which creators did bring the authentic traffic and which used some tools to create the illusion of an active and engaged audience. While this may seem obvious, it’s still worth mentioning. After the video is posted, allow 5-7 days for it to accumulate a basic number of views, then check performance metrics such as views, clicks, click-through rate (CTR), signups, and conversion rate (CR) from clicks to signups.
If you overlooked some red flags when selecting crypto channels for your launch, you might find the following outcomes: channels with high views numbers and high CTRs, demonstrating the real interest of the audience, yet with remarkably low conversion rates. In the worst-case scenario, you might witness thousands of clicks resulting in zero to just a few signups. While this might suggest technical issues in other industries, in crypto campaigns it indicates that the creator engaged in the campaign not only bought fake views and comments but also link clicks. And this happens more often than you may realize.
Summing up, choosing the right crypto creator to promote your product is indeed a tricky job that requires a lot of resources to be put into the search process.

Author
Nadia Bubennikova, Head of agency at Famesters
Fintech
Central banks and the FinTech sector unite to change global payments space

The BIS, along with seven leading central banks and a cohort of private financial firms, has embarked on an ambitious venture known as Project Agorá.
Named after the Greek word for “marketplace,” this initiative stands at the forefront of exploring the potential of tokenisation to significantly enhance the operational efficiency of the monetary system worldwide.
Central to this pioneering project are the Bank of France (on behalf of the Eurosystem), the Bank of Japan, the Bank of Korea, the Bank of Mexico, the Swiss National Bank, the Bank of England, and the Federal Reserve Bank of New York. These institutions have joined forces under the banner of Project Agorá, in partnership with an extensive assembly of private financial entities convened by the Institute of International Finance (IIF).
At the heart of Project Agorá is the pursuit of integrating tokenised commercial bank deposits with tokenised wholesale central bank money within a unified, public-private programmable financial platform. By harnessing the advanced capabilities of smart contracts and programmability, the project aspires to unlock new transactional possibilities that were previously infeasible or impractical, thereby fostering novel opportunities that could benefit businesses and consumers alike.
The collaborative effort seeks to address and surmount a variety of structural inefficiencies that currently plague cross-border payments. These challenges include disparate legal, regulatory, and technical standards; varying operating hours and time zones; and the heightened complexity associated with conducting financial integrity checks (such as anti-money laundering and customer verification procedures), which are often redundantly executed across multiple stages of a single transaction due to the involvement of several intermediaries.
As a beacon of experimental and exploratory projects, the BIS Innovation Hub is committed to delivering public goods to the global central banking community through initiatives like Project Agorá. In line with this mission, the BIS will soon issue a call for expressions of interest from private financial institutions eager to contribute to this ground-breaking project. The IIF will facilitate the involvement of private sector participants, extending an invitation to regulated financial institutions representing each of the seven aforementioned currencies to partake in this transformative endeavour.
Source: fintech.globa
The post Central banks and the FinTech sector unite to change global payments space appeared first on HIPTHER Alerts.
Fintech
TD Bank inks multi-year strategic partnership with Google Cloud

TD Bank has inked a multi-year deal with Google Cloud as it looks to streamline the development and deployment of new products and services.
The deal will see the Canadian banking group integrate the vendor’s cloud services into a wider portion of its technology solutions portfolio, a move which TD expects will enable it “to respond quickly to changing customer expectations by rolling out new features, updates, or entirely new financial products at an accelerated pace”.
This marks an expansion of the already established relationship between TD Bank and Google Cloud after the group previously adopted the vendor’s Google Kubernetes Engine (GKE) for TD Securities Automated Trading (TDSAT), the Chicago-based subsidiary of its investment banking unit, TD Securities.
TDSAT uses GKE for process automation and quantitative modelling across fixed income markets, resulting in the development of a “data-driven research platform” capable of processing large research workloads in trading.
Dan Bosman, SVP and CIO of TD Securities, claims the infrastructure has so far supported TDSAT with “compute-intensive quantitative analysis” while expanding the subsidiary’s “trading volumes and portfolio size”.
TD’s new partnership with Google Cloud will see the group attempt to replicate the same level of success across its entire portfolio.
Source: fintechfutures.com
The post TD Bank inks multi-year strategic partnership with Google Cloud appeared first on HIPTHER Alerts.
-

 Fintech5 days ago
Fintech5 days agoHow to identify authenticity in crypto influencer channels
-
Latest News4 days ago
HSBC-backed fintech Monese is considering splitting its operations as it grapples with increasing losses.
-
Latest News4 days ago
EverBank has announced a groundbreaking partnership with Finzly, poised to revolutionize payment processing.
-
Latest News4 days ago
FinTech leaders express caution regarding the promises made in #Budget2024 concerning open banking, stating that the “devil is in the details.”
-
Latest News4 days ago
Gotion High-tech’s operating profit up 391% in 2023, nearly RMB 2.8 billion invested in R&D for the year
-
Latest News4 days ago
Aurionpro Solutions acquires Arya.ai, to power next generation Enterprise AI platforms for Financial Institutions
-
Latest News5 days ago
Wells Fargo, a leading financial institution, is set to revolutionize its trade finance operations by incorporating artificial intelligence (AI) technology through its collaboration with TradeSun.
-
Latest News4 days ago
Latvian Fintech inGain Raises €650,000 for No-Code SaaS Loan Management System





