Fintech
SmallCapsDaily: Longeveron Inc. Emerges as a True Blue Bio-Tech Titan

New York, New York–(Newsfile Corp. – April 12, 2021) – Longeveron Inc. (NASDAQ: LGVN) (“Longeveron” or “Company”), a clinical stage biotechnology company developing cellular therapies for chronic aging-related and life-threatening conditions, was recently the focus of coverage by leading financial news website and publisher, SmallCapsDaily. Longeveron is swiftly becoming a leader in the biotech and regenerative medicine space, as it continues in its efforts to combat a variety of concerns and illnesses such as aging, Alzheimer’s and chronic mental health disorders by engaging in research and development of cellular therapies using stem cells.
The science behind Longeveron’s therapies, was one of the primary features in the SmallCapsDaily article; specifically, its lead candidate, Lomecel-B, a cell-based therapy obtained from medicinal signaling cells (MSCs) that are derived from the bone marrow of young healthy adult donors. Longeveron believes that by using the same cells that promote tissue repair, organ maintenance, and immune system function, it can develop safe and effective therapies for some of the most difficult disorders associated with the aging process and other medical disorders. In addition to its lead candidate, the Company’s offerings focus on a wide variety of ailments such as aging frailty, Alzheimer’s disease, related dementias (ADRD), and metabolic syndrome (MetS). Furthermore, the Company has its own manufacturing facility comprised of eight clean rooms, two research and development laboratories, and warehousing, which it uses not only for product development but also for contract manufacturing purposes.
The SmallCapsDaily coverage of Longeveron proves to be a comprehensive overview of the Company’s unique positioning within the biotech industry and the opportunity that it may represent for investors looking for stock opportunities with companies that boast a strong pipeline, a solid cash position, and clear revenue generation potential.
Key Takeaways from the SmallCapsDaily article featuring Longeveron Inc.:
- As of the first quarter of 2021, over 260 subjects have received Lomecel-B via direct injection, and there have been no serious adverse events reported that were considered related to the product candidate.
- Lomecel-B was recently approved to conduct a Phase 2 Aging Frailty clinical trial by the Japanese Pharmaceutical and Medical Devices Agency (PMDA).
- In March 2021, the company announced that the U.S. Food and Drug Administration (FDA) had granted expanded access approval for the administration of Lomecel-B to a child with HLHS (Hypoplastic Left Heart Syndrome), a rare congenital heart disease that affects about 1000 babies per year, and the full results of Longeveron’s Phase 1 trial in HLHS are expected in the second quarter of 2021.
For more insight into Longeveron Inc., and to continue reading the Small Caps Daily featured article, please click here: https://smallcapsdaily.com/longeveron-a-true-blue-biotech-titan/
Forward-looking Statements
SmallCapsDaily profiles are not a solicitation or recommendation to buy, sell or hold securities. SmallCapsDaily is a paid advertiser and is not offering securities for sale. Neither SmallCapsDaily nor its owners, operators, affiliates or anyone disseminating information on its behalf is registered as an Investment Advisor under any federal or state law and none of the information provided by SmallCapsDaily its owners, operators, affiliates or anyone disseminating information on its behalf should be construed as investment advice or investment recommendations. Small Caps Daily does not recommend that the securities profiled should be purchased, sold or held and is not liable for any investment decisions by its readers or subscribers. Information presented by Small Caps Daily may contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions, or future events or performance, are not statements of historical fact and may be “forward-looking statements.” Forward-looking statements are based on expectations, estimates and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward-looking statements may be identified through the use of words such as “expects,” “will,” “anticipates,” “estimates,” “believes,” “may,” or by statements indicating that certain actions “may,” “could,” or “might” occur.
Contact:
SmallCapsDaily
[email protected]
To view the source version of this press release, please visit https://www.newsfilecorp.com/release/80107
Powered by WPeMatico
Fintech
How to identify authenticity in crypto influencer channels

Modern brands stake on influencer marketing, with 76% of users making a purchase after seeing a product on social media.The cryptocurrency industry is no exception to this trend. However, promoting crypto products through influencer marketing can be particularly challenging. Crypto influencers pose a significant risk to a brand’s reputation and ROI due to rampant scams. Approximately 80% of channels provide fake statistics, including followers counts and engagement metrics. Additionally, this niche is characterized by high CPMs, which can increase the risk of financial loss for brands.
In this article Nadia Bubennnikova, Head of agency Famesters, will explore the most important things to look for in crypto channels to find the perfect match for influencer marketing collaborations.
-
Comments
There are several levels related to this point.
LEVEL 1
Analyze approximately 10 of the channel’s latest videos, looking through the comments to ensure they are not purchased from dubious sources. For example, such comments as “Yes sir, great video!”; “Thanks!”; “Love you man!”; “Quality content”, and others most certainly are bot-generated and should be avoided.
Just to compare:
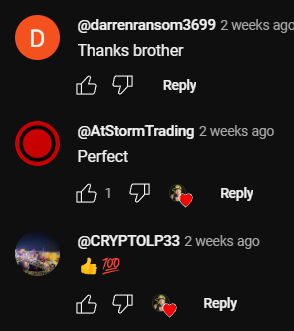
LEVEL 2
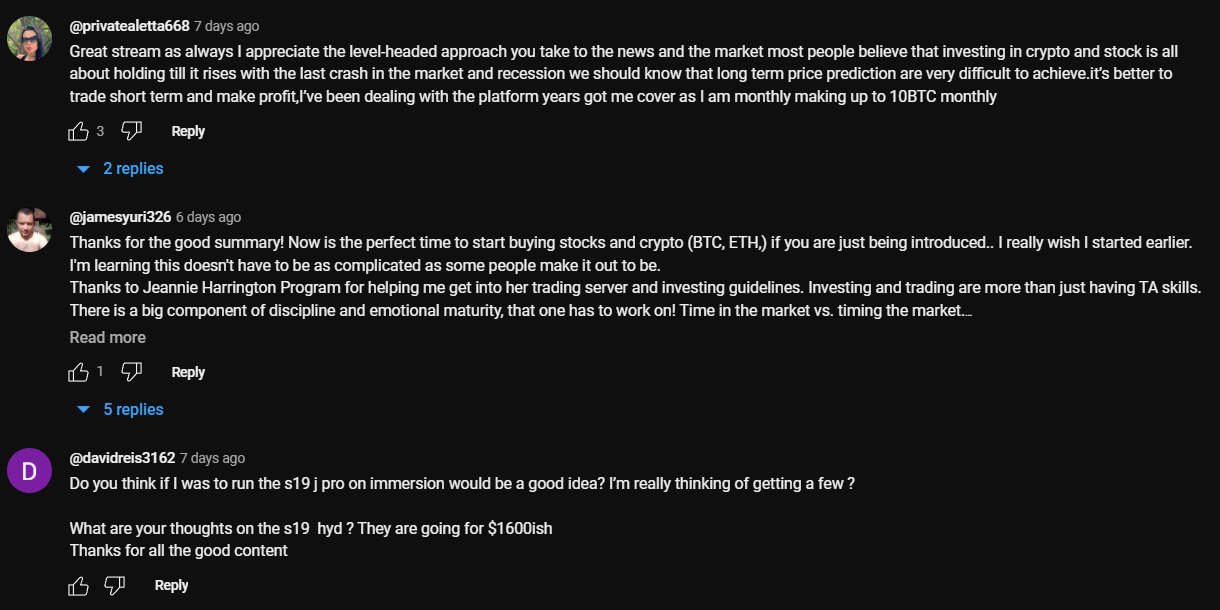
Don’t rush to conclude that you’ve discovered the perfect crypto channel just because you’ve come across some logical comments that align with the video’s topic. This may seem controversial, but it’s important to dive deeper. When you encounter a channel with logical comments, ensure that they are unique and not duplicated under the description box. Some creators are smarter than just buying comments from the first link that Google shows you when you search “buy YouTube comments”. They generate topics, provide multiple examples, or upload lists of examples, all produced by AI. You can either manually review the comments or use a script to parse all the YouTube comments into an Excel file. Then, add a formula to highlight any duplicates.
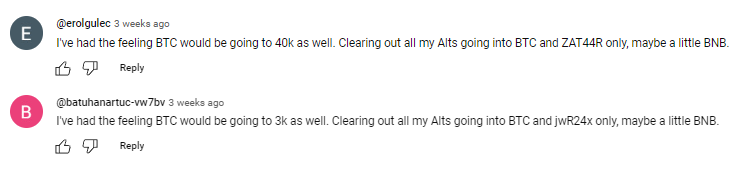
LEVEL 3
It is also a must to check the names of the profiles that leave the comments: most of the bot-generated comments are easy to track: they will all have the usernames made of random symbols and numbers, random first and last name combinations, “Habibi”, etc. No profile pictures on all comments is also a red flag.
LEVEL 4
Another important factor to consider when assessing comment authenticity is the posting date. If all the comments were posted on the same day, it’s likely that the traffic was purchased.
2. Average views number per video
This is indeed one of the key metrics to consider when selecting an influencer for collaboration, regardless of the product type. What specific factors should we focus on?
First & foremost: the views dynamics on the channel. The most desirable type of YouTube channel in terms of views is one that maintains stable viewership across all of its videos. This stability serves as proof of an active and loyal audience genuinely interested in the creator’s content, unlike channels where views vary significantly from one video to another.
Many unauthentic crypto channels not only buy YouTube comments but also invest in increasing video views to create the impression of stability. So, what exactly should we look at in terms of views? Firstly, calculate the average number of views based on the ten latest videos. Then, compare this figure to the views of the most recent videos posted within the past week. If you notice that these new videos have nearly the same number of views as those posted a month or two ago, it’s a clear red flag. Typically, a YouTube channel experiences lower views on new videos, with the number increasing organically each day as the audience engages with the content. If you see a video posted just three days ago already garnering 30k views, matching the total views of older videos, it’s a sign of fraudulent traffic purchased to create the illusion of view stability.
3. Influencer’s channel statistics
The primary statistics of interest are region and demographic split, and sometimes the device types of the viewers.
LEVEL 1
When reviewing the shared statistics, the first step is to request a video screencast instead of a simple screenshot. This is because it takes more time to organically edit a video than a screenshot, making it harder to manipulate the statistics. If the creator refuses, step two (if only screenshots are provided) is to download them and check the file’s properties on your computer. Look for details such as whether it was created with Adobe Photoshop or the color profile, typically Adobe RGB, to determine if the screenshot has been edited.
LEVEL 2
After confirming the authenticity of the stats screenshot, it’s crucial to analyze the data. For instance, if you’re examining a channel conducted in Spanish with all videos filmed in the same language, it would raise concerns to find a significant audience from countries like India or Turkey. This discrepancy, where the audience doesn’t align with regions known for speaking the language, is a red flag.
If we’re considering an English-language crypto channel, it typically suggests an international audience, as English’s global use for quality educational content on niche topics like crypto. However, certain considerations apply. For instance, if an English-speaking channel shows a significant percentage of Polish viewers (15% to 30%) without any mention of the Polish language, it could indicate fake followers and views. However, if the channel’s creator is Polish, occasionally posts videos in Polish alongside English, and receives Polish comments, it’s important not to rush to conclusions.
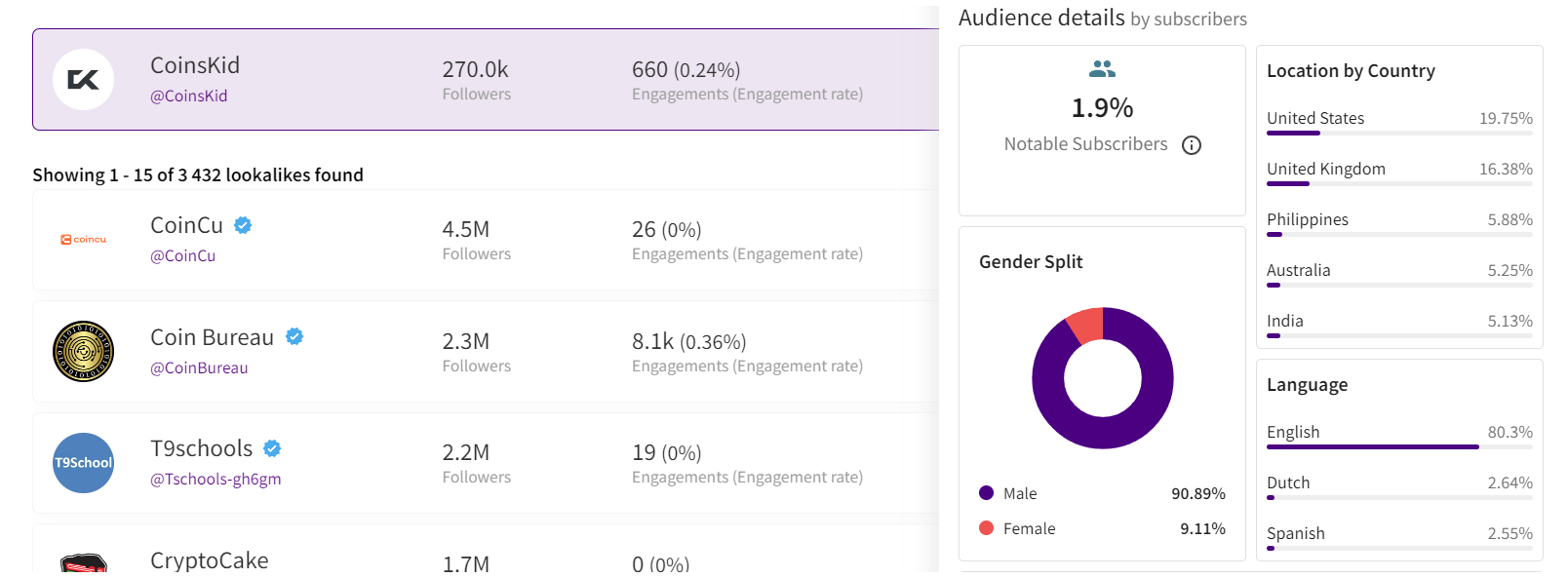 Example of statistics
Example of statistics
Wrapping up
These are the main factors to consider when selecting an influencer to promote your crypto product. Once you’ve launched the campaign, there are also some markers to show which creators did bring the authentic traffic and which used some tools to create the illusion of an active and engaged audience. While this may seem obvious, it’s still worth mentioning. After the video is posted, allow 5-7 days for it to accumulate a basic number of views, then check performance metrics such as views, clicks, click-through rate (CTR), signups, and conversion rate (CR) from clicks to signups.
If you overlooked some red flags when selecting crypto channels for your launch, you might find the following outcomes: channels with high views numbers and high CTRs, demonstrating the real interest of the audience, yet with remarkably low conversion rates. In the worst-case scenario, you might witness thousands of clicks resulting in zero to just a few signups. While this might suggest technical issues in other industries, in crypto campaigns it indicates that the creator engaged in the campaign not only bought fake views and comments but also link clicks. And this happens more often than you may realize.
Summing up, choosing the right crypto creator to promote your product is indeed a tricky job that requires a lot of resources to be put into the search process.

Author
Nadia Bubennikova, Head of agency at Famesters
Fintech
Central banks and the FinTech sector unite to change global payments space

The BIS, along with seven leading central banks and a cohort of private financial firms, has embarked on an ambitious venture known as Project Agorá.
Named after the Greek word for “marketplace,” this initiative stands at the forefront of exploring the potential of tokenisation to significantly enhance the operational efficiency of the monetary system worldwide.
Central to this pioneering project are the Bank of France (on behalf of the Eurosystem), the Bank of Japan, the Bank of Korea, the Bank of Mexico, the Swiss National Bank, the Bank of England, and the Federal Reserve Bank of New York. These institutions have joined forces under the banner of Project Agorá, in partnership with an extensive assembly of private financial entities convened by the Institute of International Finance (IIF).
At the heart of Project Agorá is the pursuit of integrating tokenised commercial bank deposits with tokenised wholesale central bank money within a unified, public-private programmable financial platform. By harnessing the advanced capabilities of smart contracts and programmability, the project aspires to unlock new transactional possibilities that were previously infeasible or impractical, thereby fostering novel opportunities that could benefit businesses and consumers alike.
The collaborative effort seeks to address and surmount a variety of structural inefficiencies that currently plague cross-border payments. These challenges include disparate legal, regulatory, and technical standards; varying operating hours and time zones; and the heightened complexity associated with conducting financial integrity checks (such as anti-money laundering and customer verification procedures), which are often redundantly executed across multiple stages of a single transaction due to the involvement of several intermediaries.
As a beacon of experimental and exploratory projects, the BIS Innovation Hub is committed to delivering public goods to the global central banking community through initiatives like Project Agorá. In line with this mission, the BIS will soon issue a call for expressions of interest from private financial institutions eager to contribute to this ground-breaking project. The IIF will facilitate the involvement of private sector participants, extending an invitation to regulated financial institutions representing each of the seven aforementioned currencies to partake in this transformative endeavour.
Source: fintech.globa
The post Central banks and the FinTech sector unite to change global payments space appeared first on HIPTHER Alerts.
Fintech
TD Bank inks multi-year strategic partnership with Google Cloud

TD Bank has inked a multi-year deal with Google Cloud as it looks to streamline the development and deployment of new products and services.
The deal will see the Canadian banking group integrate the vendor’s cloud services into a wider portion of its technology solutions portfolio, a move which TD expects will enable it “to respond quickly to changing customer expectations by rolling out new features, updates, or entirely new financial products at an accelerated pace”.
This marks an expansion of the already established relationship between TD Bank and Google Cloud after the group previously adopted the vendor’s Google Kubernetes Engine (GKE) for TD Securities Automated Trading (TDSAT), the Chicago-based subsidiary of its investment banking unit, TD Securities.
TDSAT uses GKE for process automation and quantitative modelling across fixed income markets, resulting in the development of a “data-driven research platform” capable of processing large research workloads in trading.
Dan Bosman, SVP and CIO of TD Securities, claims the infrastructure has so far supported TDSAT with “compute-intensive quantitative analysis” while expanding the subsidiary’s “trading volumes and portfolio size”.
TD’s new partnership with Google Cloud will see the group attempt to replicate the same level of success across its entire portfolio.
Source: fintechfutures.com
The post TD Bank inks multi-year strategic partnership with Google Cloud appeared first on HIPTHER Alerts.
-
Latest News7 days ago
HSBC-backed fintech Monese is considering splitting its operations as it grapples with increasing losses.
-
Latest News7 days ago
EverBank has announced a groundbreaking partnership with Finzly, poised to revolutionize payment processing.
-
Latest News7 days ago
FinTech leaders express caution regarding the promises made in #Budget2024 concerning open banking, stating that the “devil is in the details.”
-
Latest News6 days ago
Aurionpro Solutions acquires Arya.ai, to power next generation Enterprise AI platforms for Financial Institutions
-
Latest News7 days ago
Gotion High-tech’s operating profit up 391% in 2023, nearly RMB 2.8 billion invested in R&D for the year
-
Latest News7 days ago
Latvian Fintech inGain Raises €650,000 for No-Code SaaS Loan Management System
-
Latest News7 days ago
Credit card fintech Pliant lands €18m Series A extension led by PayPal Ventures
-
Latest News7 days ago
Fintech Powerhouse CRED Receives In-Principle Approval for Payment Aggregator License



